Building on the outcomes and recommendations of recent regional webinars on (a) influenza surveillance and (b) insights on both the northern and southern hemisphere vaccine composition meetings, the WHO Regional Office for South‑East Asia (SEARO), in collaboration with Global Influenza Programme (GIP) of WHO headquarters , conducted a two‑day regional virtual training on “Data Uploading in RespiMart and Introduction to Excel Power Query” on 21–22 April 2026.
The training was delivered as a direct follow‑up and build-on activity, translating regional discussions into practical action to address identified gaps in the quality, completeness, and timeliness of influenza data reporting across Member States.
During the earlier webinars, Member States highlighted several recurring challenges affecting influenza data reporting, including:
- Data cleaning and standardization issues, leading to inconsistencies across datasets.
- Difficulties in integrating epidemiological and laboratory data, limiting comprehensive analysis.
- Errors and delays in reporting to the global platform, affecting data timeliness and usability.
In response, the training was designed as a hands‑on technical intervention to operationalize these recommendations and strengthen routine influenza data management and reporting workflows across Member States.
This training contributes directly to the implementation of the WHO Global Influenza Strategy (2019–2030) and supports the High-Level Implementation Pla -III of the Pandemic Influenza Preparedness (PIP) Partnerships Contributions, particularly its collaborative surveillance through the Global Influenza Surveillance and Response System (GISRS) output, by strengthening country capacity for timely, high‑quality influenza data reporting.
By strengthening data reporting systems such as RespiMart, countries are better equipped to:
- Improve the accuracy, completeness, and timeliness of influenza surveillance data, strengthening overall data quality for national and global use
- Reduce data validation errors and reporting gaps, ensuring more consistent and reliable submissions to global platforms
- Support timely risk assessment and evidence‑based decision‑making, enabling more effective public health action at national, regional, and global levels
The training brought together National Influenza Centre (NIC) focal points, data managers, surveillance officers, and reporting staff from countries across the WHO South‑East Asia Region.
Key focus areas of the training included:
- Data cleaning, transformation, and merging using Excel Power Query, to enable efficient preparation of surveillance datasets
- Step‑by‑step guidance on RespiMart data upload and validation procedures, supporting accurate and timely reporting
- Identification and resolution of common data reporting challenges to ensure consistent data reporting by countries in the WHO Southeast Asia Region.
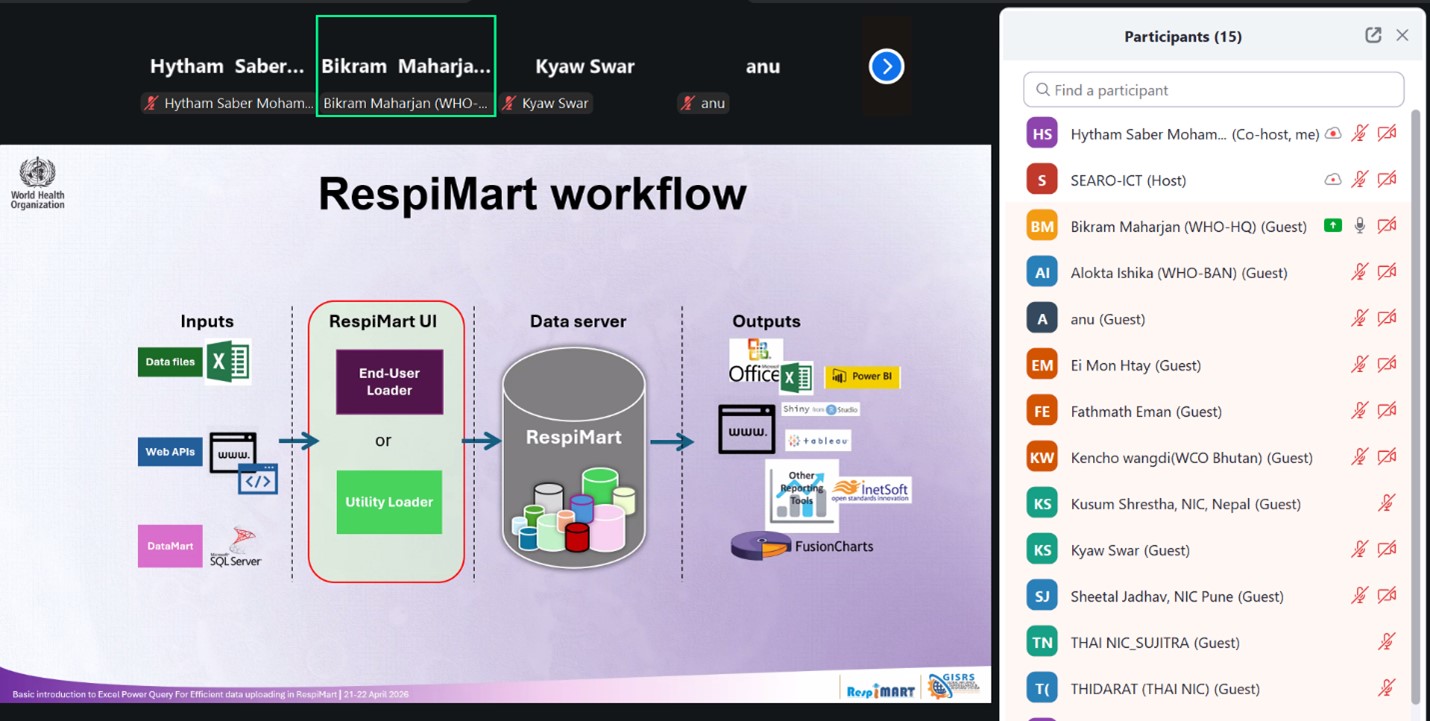
Figure 1. RespiMart workflow, illustrating the core processes underpinning strengthened influenza data reporting systems that support collaborative surveillance and timely public health decision‑making.
From a technical perspective, the training emphasized the importance of standardization and interoperability of surveillance datasets across epidemiological and laboratory data streams. Participants were guided on structuring datasets using unique identifiers (such as patient or sample IDs) to enable accurate data merging, applying data validation rules aligned with RespiMart templates, and systematically addressing common inconsistencies. These included missing denominators, incorrect or non‑standard date formats, and duplicate records, all of which are critical to ensuring data quality, comparability, and reliable analysis.
The use of Excel Power Query enabled automated workflows for data transformation, including data type harmonization, error handling, and aggregation (such as weekly reporting). This automation helped reduce manual processing errors and improve the reproducibility of routine data management processes. By supporting more robust and standardized data pipelines, this approach ensures that submitted datasets are consistent with WHO reporting standards and readily usable for regional and global influenza analysis and interpretation.
 |  |
Figure 2. Live demonstration of data cleaning and transformation processes using Excel Power Query conducted during the training
Participants were also reminded of key considerations to safeguard the integrity of the reporting process. These included verifying data completeness prior to submission, ensuring alignment and consistency between epidemiological and laboratory datasets, and carefully reviewing validation reports generated by RespiMart before finalizing and committing data.
Particular attention should be given to avoiding inconsistent reporting periods, duplicate entries, and incomplete denominators, as these issues can significantly affect data interpretation at regional and global levels. Adherence to established reporting timelines and standard operating procedures is therefore critical to maintaining data reliability, consistency, and comparability across countries.
This initiative reflects a progressive, build‑on approach to capacity strengthening, in which regional dialogue is systematically translated into targeted technical support and practical training. It also underscores the importance of sustained collaboration between Member States and three levels of WHO in strengthening influenza surveillance systems and enhancing data‑driven public health action across the Region.
Participants are expected to apply the knowledge and skills gained through this training in their routine work and to further disseminate these good practices within their respective teams and institutions. As countries continue to strengthen integrated influenza and other respiratory pathogen surveillance systems, such follow‑up activities play a critical role in ensuring that regional recommendations are translated into tangible and sustained improvements, thereby reinforcing the foundation for effective preparedness, early detection, and response to influenza and other respiratory threats.
