Procurement and technical specifications
There is a need for advice on technical specifications of medical devices in many settings. Decision makers should consider technical specifications before purchase or donation. Technical specifications improve access to high quality, safe and efficacious medical devices. They allow for adequate planning of implementation, functioning and decommissioning of medical devices. Technical specifications facilitate the management of financial, infrastructural and human resources.
Therefore, WHO has developed some technical specifications for specific populations (e.g. newborns) and needs (e.g. oxygen). They are free to download on this page. WHO technical specification also provide guidelines in procurement and acquisition of medical devices.
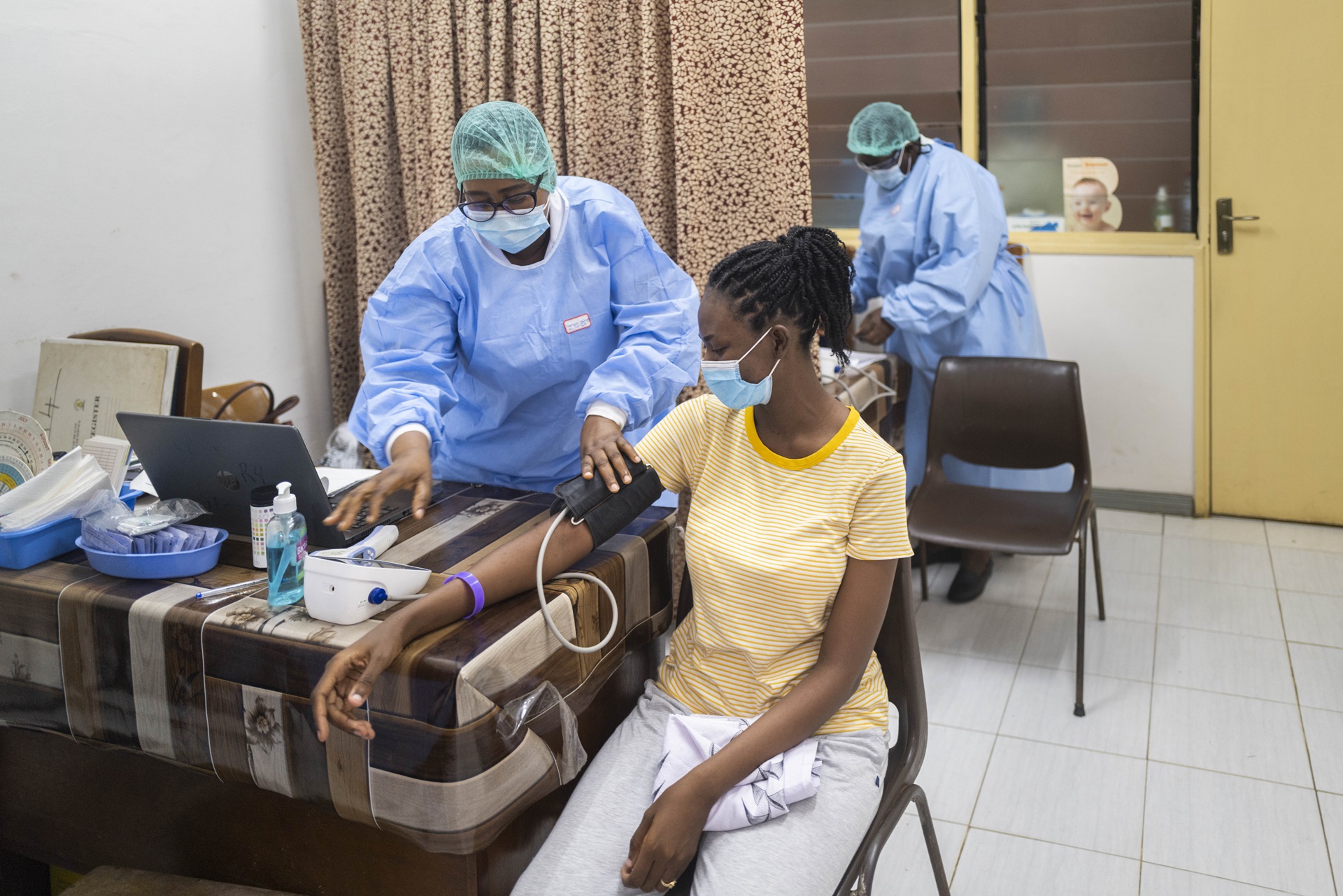
Midwife Rebecca Kpabitey Maame checks the blood pressure of a patient, who delivered her baby Ishod at Ridge Hospital and is now bringing him for a postnatal visit.
WHO procurement guidance
WHO technical specifications of medical devices
WHO technical specifications package
WHO developed the technical specifications package since 2014. An updated electronic version is being developed.

WHO Technical Specifications (TS) are intended for technical health workers and health stakeholders such as:biomedical engineershospital managersplanning...

Six million women and children could be saved by 2017, if they had access to simple, essential and affordable medicines, medical devices and health supplies....

The WHO technical specification for 61 medical devices was developed in accordance with essential medical devices needed for health care facilities. It...

WHO technical specifications for blood glucose meter
The WHO technical specification for blood glucose meter was developed by WHO in collaboration with a working group of experts. The technical specifications...
Global collaborations for medical devices
WHO directs and coordinates international health work through collaboration. WHO partners with countries, the United Nations system, international organizations, civil society, foundations, academia, and research institutions. Official collaborations with WHO are conducted in the following forms:
Contact
